Revolutionizing Neuroscience with our dry electrode technology
Electroencephalography (EEG) is the measurement of electric potentials at the scalp due to currents flowing through scalp tissue. The strength and distribution of currents (and therefore potentials) reflects the intensity and position of activity in the underlying neural tissue. EEG signal is measured between two electrodes, the position of which determines the recorded brain area. Multiple electrodes are typically placed in standard arrangements that cover the entire scalp and allow investigators to observe the activity of the entire brain simultaneously. EEG is typically recorded as a time-series of potential differences, which can be evaluated visually, or analyzed spectrally, or through the use of source localization methods. The principal spectral components of EEG are divided into the following signal bands: delta (0-4 Hz), theta (4-8 Hz), alpha (8-12 Hz), beta (above 12 Hz) and gamma (above 40 Hz). Many studies have related changes in various spectral components of EEG to specific cognitive functions and clinical conditions.
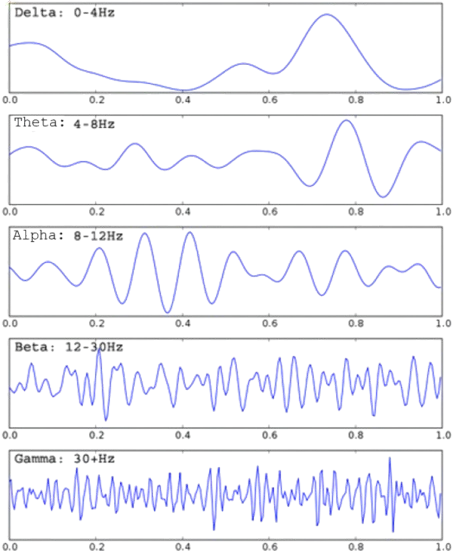

In clinical settings, continuous EEG recordings are used for monitoring sleep and anesthesia, and diagnosis of epilepsy, coma, brain death, and more recently ADHD. In research environments, EEG recordings are used in a wide range of applications, including neuroscience and cognitive psychology research into understanding brain function; Brain-Computer Interfaces to allow control of computers or machines from neural signals; neurofeedback where users are able to train their brains to generate specific activity patterns; neuromarketing where marketing companies seek to tap directly into brain signals to assess subjects’ engagement levels; neuroergonomics where researchers quantify mental workload under various strains; gaming where EEG signals are used to control computer games and toys; etc.EEG is typically recorded as a time series of potential differences, which can be evaluated visually, or analyzed spectrally, or through the use of source localization methods. The principal spectral components of EEG are divided into the following signal bands: delta (0-4 Hz), theta (4-8 Hz), alpha (8-12 Hz), beta (above 12 Hz) and gamma (above 40 Hz). Many studies have related changes in various spectral components of EEG to specific cognitive functions and clinical conditions.
Conventional EEG systems require highly trained technicians to abrade the scalp and apply electrodes and conductive gels. The process is time consuming, irritating to users, and prone to noise and artifacts, thereby limiting the practical usability of EEG monitoring in applied environments. QUASAR’s dry electrode EEG sensors overcome this technical hurdle by eliminating the need for abrasive and conductive gels EEG and enabling rapid and minimally intrusive recordings in naturalistic environments.
This uniquely high fidelity yet practical EEG recording technology builds on dry electrode sensors developed by QUASAR under funding from DARPA, the Army, the Air Force, the NSF and the NIH, and integrated into headset designs that allow use by minimally trained personnel without assistance. These ultra-high impedance dry sensors record high quality EEG through hair without the need for skin preparation of any kind. (Figure 1A) QUASAR’s dry sensors rely on several technological advances for their performance, including patented circuit design, active cancellation of common mode electrical artifacts (such as triboelectric discharges or 60Hz mains) for reduction of artifacts and environmental noise, and shielding to reduce sensitivity to electrostatic noise. 1,2,3 Furthermore, the mechanical structure of the headset includes a nested design of spring-isolated pods that ensures electrode stability and motion isolation, allowing artifact-free recordings during ambulation. (Figure 1B) The system features a fast and easy setup, designed to be worn like a baseball cap, automatically positioning 21 sensors and enabling EEG measurements in under 5 minutes. With ultra-high impedance dry sensors, it provides high-fidelity recordings through hair, without requiring skin preparation. Its patented mechanical isolation design stabilizes electrodes for artifact-free recordings, even during movement, and built-in active noise cancellation reduces environmental interference, such as 60Hz mains and electrostatic noise. Developed in collaboration with Wearable Sensing and QUASAR, it connects users to a broad network of academic and industry partners, supporting diverse research applications. Manufactured in a US-based, ISO 13485-certified facility, the system ensures consistent, high-quality standards.


Signal Quality: For conventional passive wet electrode EEG, the skin is abraded and conductive gel is applied between the skin and electrode to provide a low impedance contact. This low impedance is critical to ensure good signal quality for these passive systems, as otherwise, the electrode tip and cables would pick up stay electromagnetic interference (EMI) from the environment, which could mask the relatively small EEG signals. Accordingly, passive wet electrode systems typically require contact impedances below 10kOhms for optimal performance, while active wet electrode systems that have amplifiers at the electrode tip require impedances below 50kOhms, as they amplify the EEG signal at the source, and thus reduce the impact of long wires on EMI pick-up. QUASAR’s active dry electrodes’ ultra-high input impedance amplifiers (>47 GOhms) and electronics are well matched to handle contact impedances up to 1-2 MOhms. The signal quality recorded from these dry sensors is comparable to that obtained from wet electrodes attached by a technician. (Figure 2B).
Figure 2. Comparison of EEG signals recorded using QUASAR (red) and wet (blue) electrodes simultaneously: A) Alpha activity obtained by asking subject to close eyes, EEG signal is filtered between 1 Hz and 30 Hz (B) Auditory Event-Related Potential (ERP) generated by averaging EEG responses over 200 tone stimulations and filtering between 0.5 Hz and 40 Hz. In both cases, signal correlations between wet and dry electrode recordings are > 90%.
Figure 2A&B show, respectively, an overlay of alpha activity and an auditory evoked potential simultaneously recorded with juxtaposed wet and dry electrodes; these data had 90% correlation between wet and dry electrodes.
Furthermore, to improve the dry electrodes’ resistance to EMI, the QUASAR sensors’ electrode tips and pre-amplifiers are encased in individual Faraday cages that protect them from ambient artifacts from all sides, except the side where the electrode tip makes contact with the head.
(Figure 3A) This combination of ultra-high impedance electronics and Faraday shielding provides a different and superior solution to protecting the EEG signals from ambient EMI than skin abrasion and gel application. Figure 3B shows a power spectral density (PSD) of EEG signals of an infant having a 3Hz seizure simultaneously recorded with wet and dry electrode systems. 6 The signals had an 84% correlation, with the difference being attributed to the spatial distance between the electrodes relative to the small infant’s head. However, it is noteworthy that the medical-grade passive wet electrode system picked up six times more 60 Hz mains noise than QUASAR’s shielded active dry electrode system. (Figure 3B)
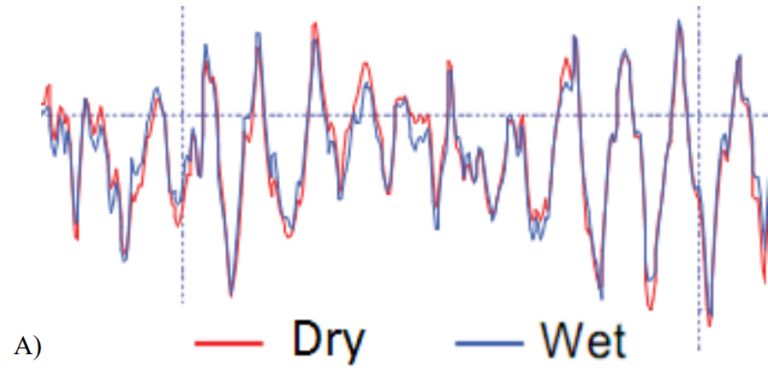
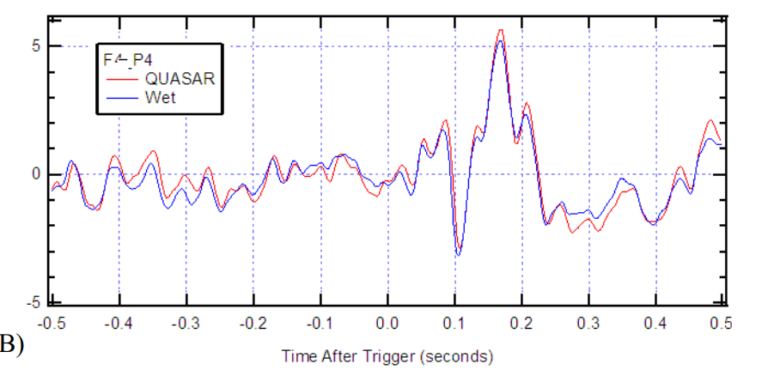
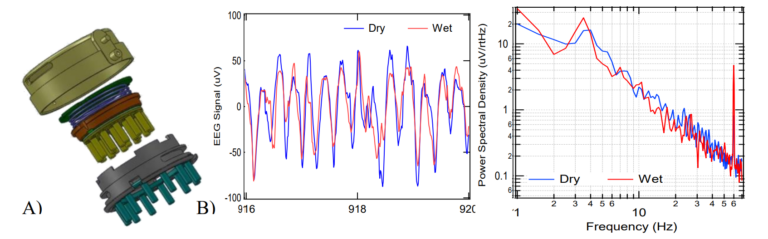
Figure 3. Artifact resistance: A) Illustration of QUASAR’s dry electrode showing the silver/silver chloride electrode tip pins in yellow; immediately behind the electrode is a green printed circuit board (PCB) that has the ultra-high impedance amplifiers and QUASAR’s custom circuitry with active shielding, the electronics are encased in a resin filled enclosure (red); the electronics are spring-loaded to protect against motion artifacts, and are housed inside a Faraday cage (grey enclosure with green pins and its backside is the larger green PCB above the purple spring). B) EEG trace and Power Spectral Density (PSD) graph of EEG signals recorded using QUASAR (blue) and wet (red) electrodes simultaneously. This signal was recorded from an infant who was having a seizure (3 Hz peak). The wet and dry spectra look similar except for the 60Hz mains frequency spike that is 8 times larger on the medical grade passive wet electrode system. QUASAR’s dry electrode systems have been evaluated by numerous groups since 2009. In 2009, Air Force researchers evaluated QUASAR’s EEG system on 19 subjects and reported “results confirm that the data collected by the new [QUASAR] system is comparable to conventional wet technology”. This dry electrode technology was validated in Brain Computer Interface paradigms and found to enable comparable classifications to those obtained with wet electrode recordings. 8 Furthermore, researchers and epileptologists at the Texas Comprehensive Epilepsy Program of the University of Texas Health Science Center at Houston (UTHSC), conducted a clinical evaluation of QUASAR’s EEG system, and determined that data quality is suitable for diagnosing status epilepticus and seizure activity. More recently, Mahdid et al reported indistinguishable results between spectral and topographic analyses between the DSI-24 and a gold-standard wet electrode system (EGI) as well as comparable phase lead/lag relationships and regions of dominant functional connectivity. Most importantly this technology is now routinely being used in research such as developing biomarkers of mild traumatic brain injury (mTBI) , and hypoxia.
Simultaneous measurements of ERP signals using dry electrode and wet electrodes excellent conservation of signal morphology between signals obtained from wet and dry electrodes; both in the pre-trigger “noise” segment, and in the N100-P200 ERP component. This is evident both in a visual inspection of the traces presented in the illustrative Figures, and also by the fact that the correlation values exceed 90% for both anterior-posterior ERP signals and that the SNRs for both electrode technologies are equivalent.
In order to be adopted and used in an applied environment, the monitoring system should be easy to use, comfortable and not interfere with task performance. Accordingly, QUASAR’s headset utilizes a patented combination of spring-loaded arms and sensor mounts to allow it to be put rapidly on the head in the manner of a baseball cap, and controlled expansion joints automatically position 21 sensors simultaneously according the International 10/20 System over a wide range of head sizes. 4 During testing of the headset, the time to don the headset and begin EEG measurements was less than 5 minutes.
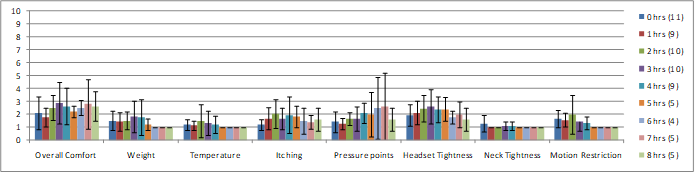
Importantly, these systems are also very comfortable. During a series of 4 or 8-hours long recording experiments, QUASAR surveyed the subjects on the comfort of the dry sensor EEG headset designed for seated environments. Subjects reported comfort on a category rating scale from 1 – 10, where 1 relates to “unnoticeable”, 5 “noticeable but comfortable”, and 10 “intolerable.” Figure 3 presents a summary of the survey data divided into 8 specific categories. Results indicate that on all categories the subjects reported high tolerance and comfort, and even after 8 hours of wear, subjects reported the headset was comfortable on all categorical measures. (subject drop-out is due to experiment end, not discomfort)
In summary, QUASAR’s dry sensor EEG technology is thus ideally suited for use in applications real-world training environments, both for its high fidelity, artifact-free signal quality, and for its practical, comfortable wireless headset that does not interfere with task performance. This technology is uniquely ready for practical use in training environments.
Wearable Sensing has licensed QUASAR’s technology for commercializing its applications.